Turning a concept into a thriving medical device business requires more than innovation. It demands strategy, regulatory expertise, clinical insight, and market intelligence. At Simplimedica, we offer specialized Medical Device Startups Service, helping innovators transform ideas into successful, market-ready products.
With over 25 years of combined experience in medical device regulations, clinical evaluation, market strategy, and funding pathways, Simplimedica provides early-stage companies with the tools, guidance, and connections needed to navigate the complex healthcare landscape.

For any medical device start-up, understanding the market is crucial. Our Medical Device Startups Service includes detailed market analysis and competitive intelligence to inform your strategic decisions.
We evaluate market potential, adoption rates, and revenue opportunities. This allows start-ups to prioritize target regions, estimate sales projections, and make informed investment decisions. Accurate forecasting ensures resources are allocated efficiently and supports investor confidence.
Simplimedica identifies key decision-makers across the healthcare ecosystem, from hospital procurement teams and clinicians to investors and payers. Effective stakeholder mapping ensures your start-up engages the right partners for clinical adoption, funding, and market expansion.
Understanding the competitive landscape is essential. Our Medical Device Startups Service includes analyzing competitor products, pricing strategies, clinical evidence, and market positioning. This helps start-ups identify gaps, differentiate offerings, and craft compelling value propositions.
A strategic go-to-market plan is key to a successful medical device launch. Simplimedica’s services for early-stage innovators include evaluating healthcare infrastructure, aligning product development with CE, UKCA, and FDA regulations, and creating market entry roadmaps with timelines for commercial launch. Our tailored strategies give start-ups the clarity and confidence needed to scale their devices efficiently.
Clinical trials can be a key challenge for early-stage medical device companies. Simplimedica’s Medical Device Startups Service offers guidance on trial strategy, site selection, and study design, including identifying research institutions in the UK, GCC, and USA. We design trials to balance compliance, cost, and efficiency, supporting feasibility studies and pivotal trials to generate credible data for regulatory submissions, investor pitches, and payer engagement.
Real-world evidence is crucial for product adoption, reimbursement, and regulatory compliance. Simplimedica’s Medical Device Startups Service supports by designing post-market studies, conducting health economic assessments, and collecting patient-reported outcomes. These services help generate valuable clinical and economic evidence to support regulatory submissions and commercialization efforts.

25+ Years of Regulatory Experience

Simplimedica provides specialized Medical Device Startups Service, giving early-stage innovators a competitive advantage. Our expertise includes:
We integrate regulatory, clinical, and market expertise, ensuring start-ups have a clear path from concept to commercialization.
A clear regulatory strategy is essential for start-up success. Simplimedica develops comprehensive Regulatory Roadmaps as part of our Medical Device Startups Service.
A well-structured regulatory roadmap not only ensures compliance but also builds investor confidence, demonstrating a clear path to market entry and commercial growth.g
Securing funding is often a significant challenge for medical device start-ups. Simplimedica’s Medical Device Startups Service includes assistance in accessing grants, venture capital, and other funding opportunities.
Expertise in Innovate UK, NIHR, Horizon Europe, and Biomedical Catalyst programs, with guidance on proposal development, submission, and post-award management.
Support for NIH, NSF, BARDA, SBIR/STTR, and other federal funding programs, including assistance with regulatory and reporting requirements.
Identification of regional funding programs and government-backed healthcare initiatives, with end-to-end support from application to compliance reporting.
Craft and submit competitive grant applications tailored to funder priorities.
Ensure compliance, reporting, and proper fund utilization.
This ensures start-ups have access to funding streams that accelerate product development, regulatory approvals, and market entry.
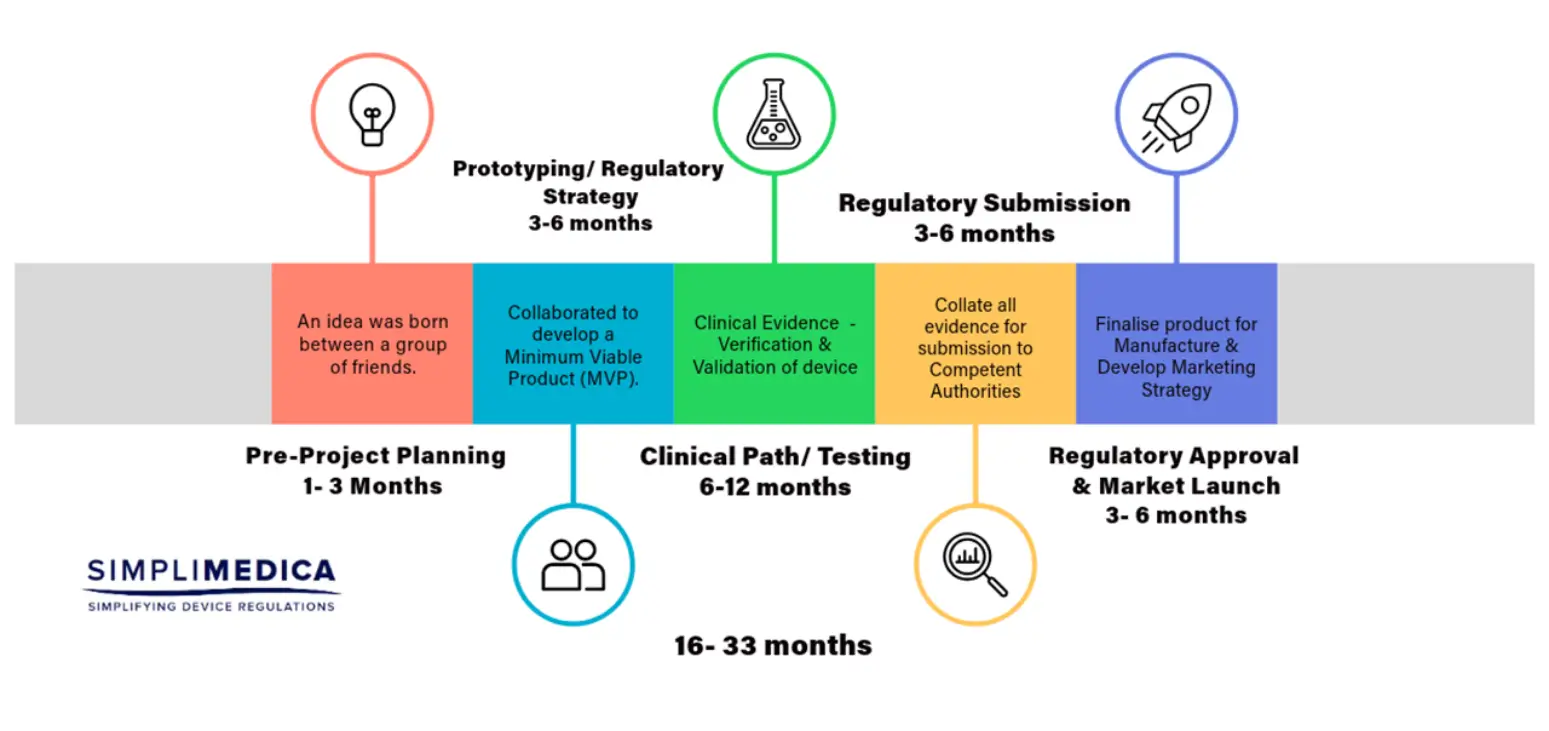

Medical device start-ups progress through distinct funding stages. Simplimedica helps align funding strategies with regulatory milestones.
Pre-Seed Stage (1–2 years)
Seed Stage (1–2 years)
Growth Stage
Simplimedica’s Medical Device Startups Service equips early-stage innovators with the guidance, resources, and strategic insight required to succeed.
Book a free consultation to:
With our support, medical device start-ups can transform innovative ideas into market-ready solutions, ensuring compliance, patient safety, and sustainable growth.
